Acid and Base Equilibria Electrolytes Strong Conduct electricity Weak Poor conductors of electricity Nonelectrolytes Do not conduct electricity. - ppt download

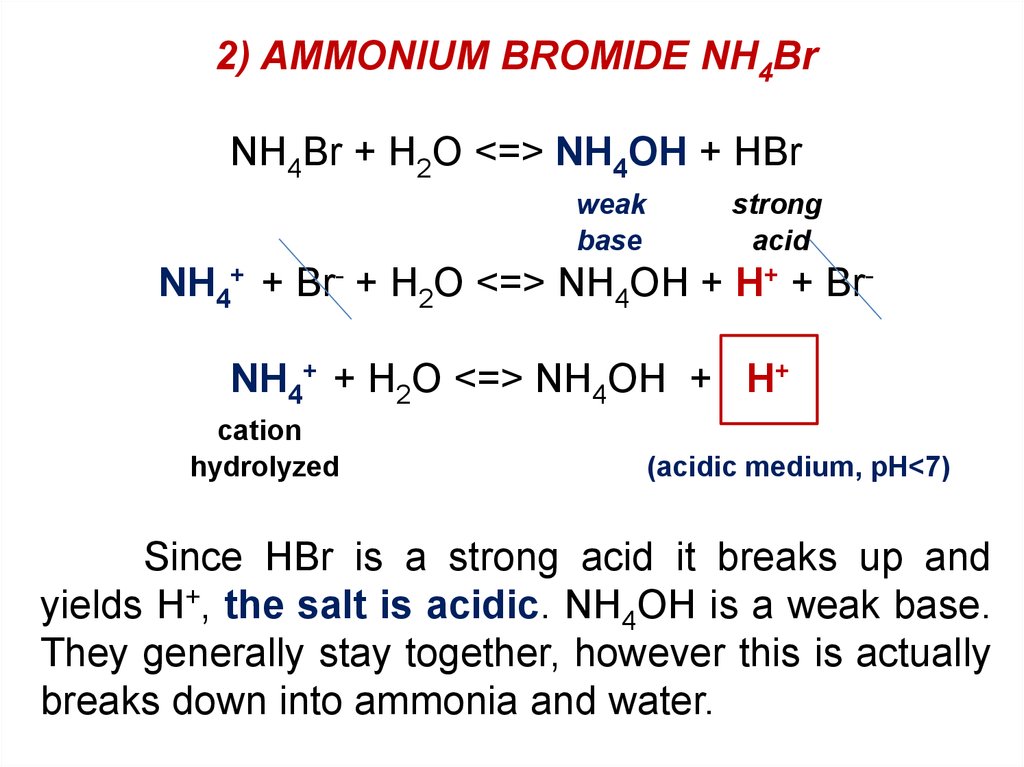
NH4Br --> acid LiNO3 --> neutral K2SO3 --> basic KCl --> neutral Na2S --> basic - Home Work Help - Learn CBSE Forum

Chapt 12 Practice problems soln - Introductory Chemistry 030 (Prof. J. R. Tolman) Acids and Bases - Studocu

SOLVED: 1. Answer and show all work: a. Are solutions of ammonium bromide, NH4Br; acidic, neutral or basic? Explain, using a chemical equation to demonstate your point. b. Explain hos sodium bicarbonate,