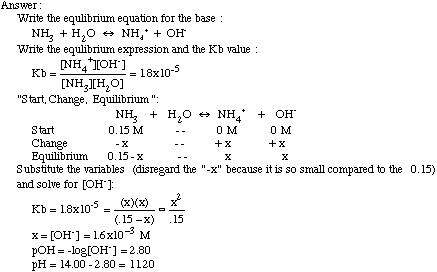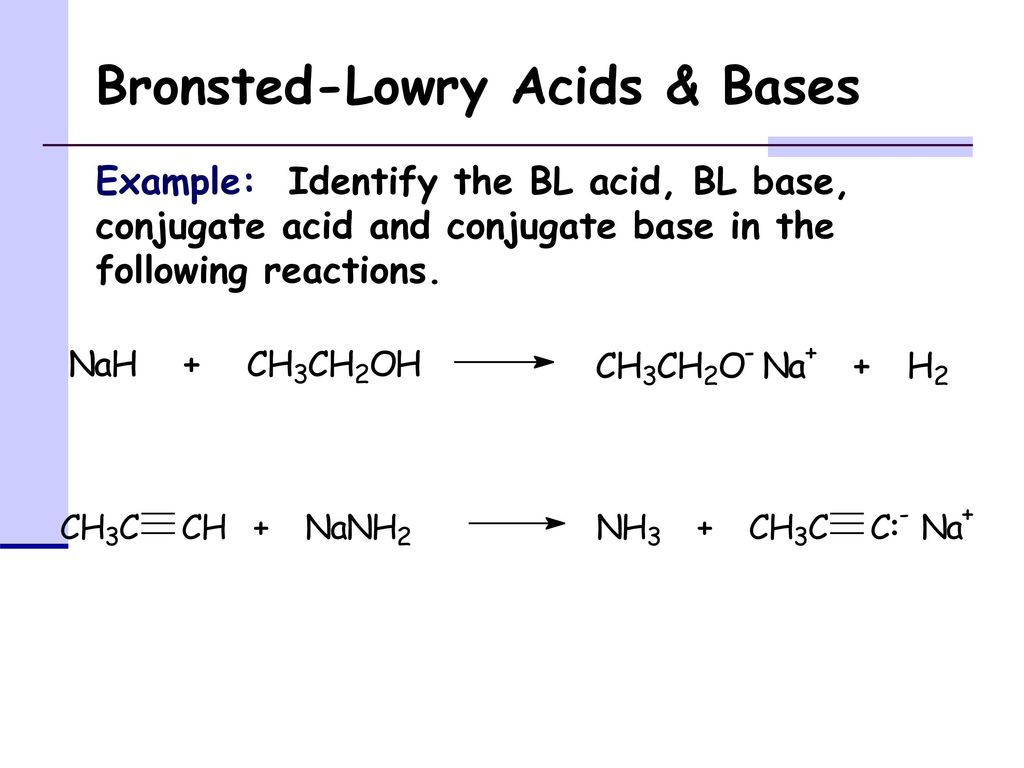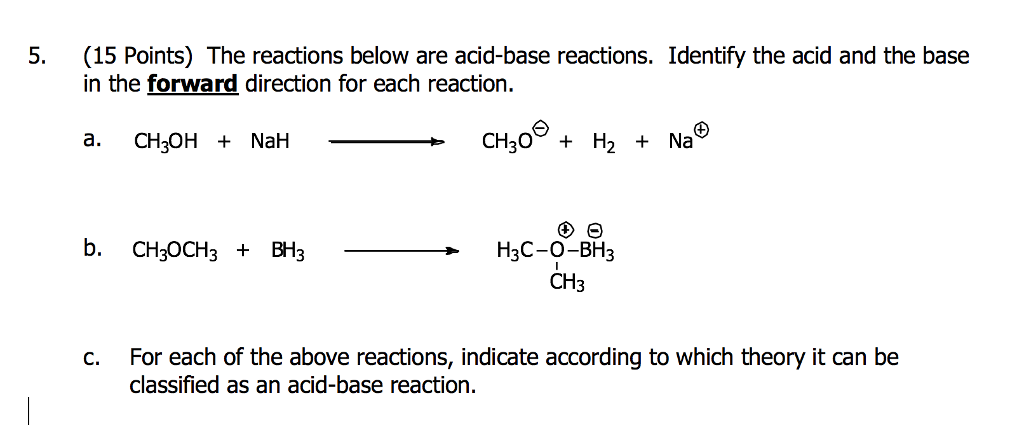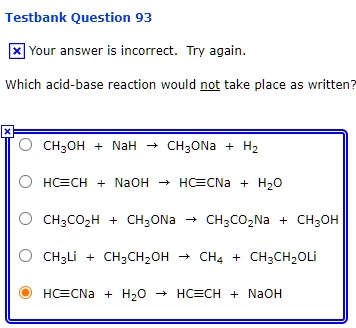
SOLVED: Testbank Question 93 Your answer incorrect Try again Which acid base reaction would not take place as written CHzOH Nah CHzONa HCCH NaOH HCCNa Hzo CH;COzH CHzONa CH;COzNa CHzOH CHzLi CHzCHzOH

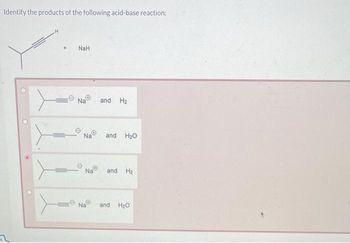
What product is formed when the following compound is treated with NaH? The following acid-base reactions were a step in a synthesis of a commercially available drug. | Homework.Study.com
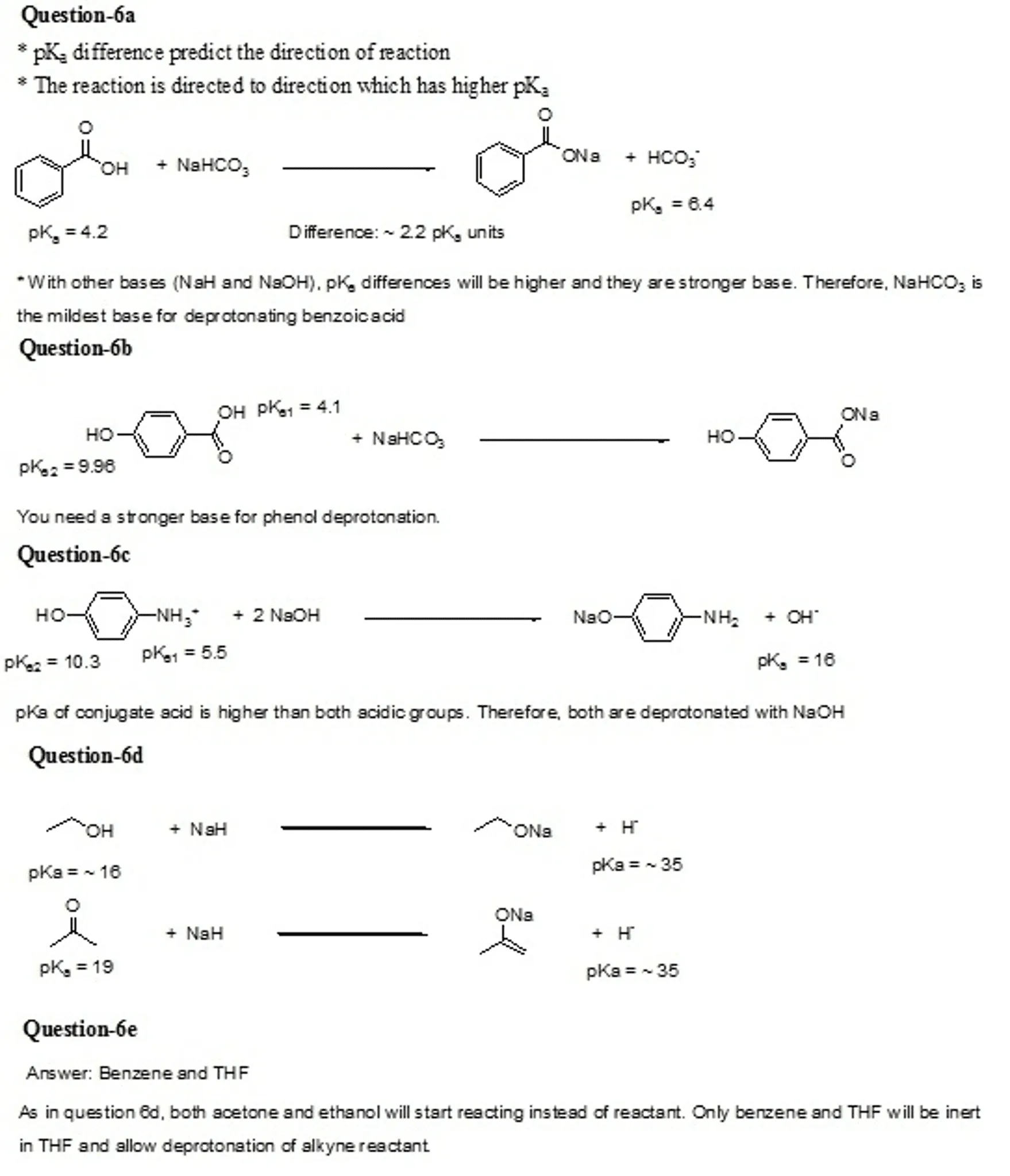
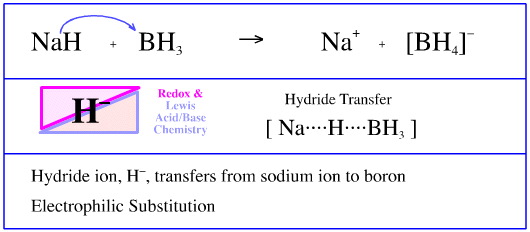
OneClass: The pKa of the conjugate acid of sodium hydride (NaH) is about 35, the pKa of sodium hydrox...

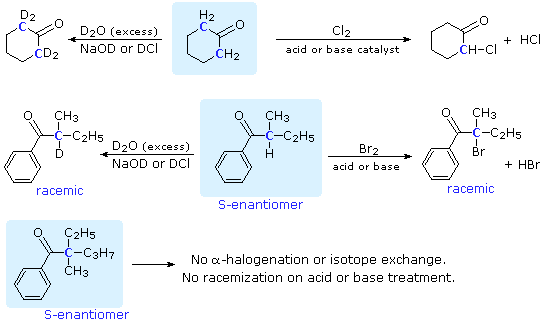
SOLVED: Sodium hydride (NaH) is convenient source Of the hydride anion (H ) the conjugate base of Hz: In contrast t0 other hydride reagents that we will learn about next semester; NaH

What product is formed when the given compound is treated with NaH? The given acid-base reactions were a step in a synthesis of a commercially available drug. | Homework.Study.com

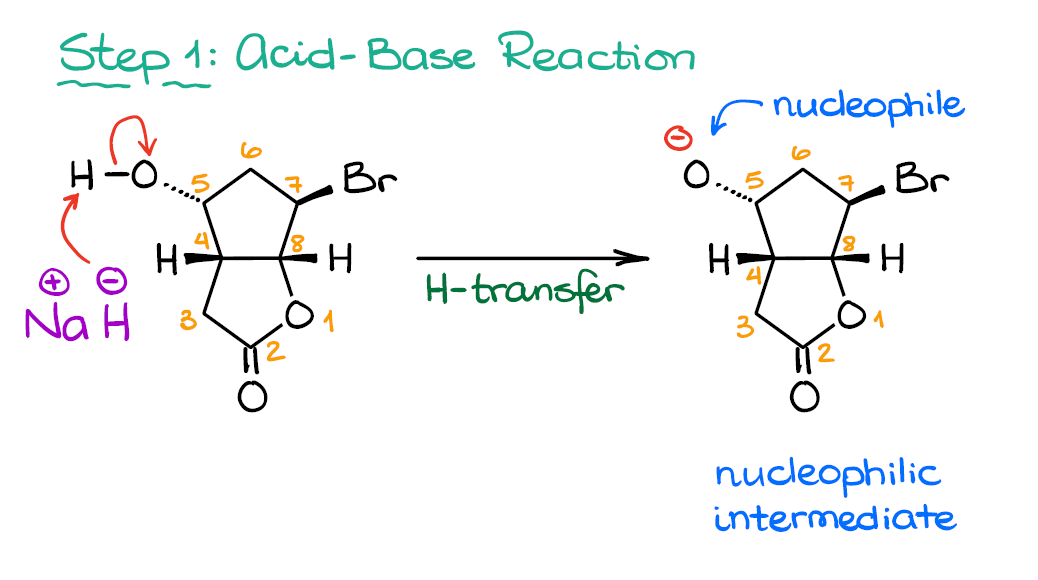
OneClass: Predict the structures of BOTH bronsted acid base reaction of NaH with typical thiol. What ...